What is a Buffer Solution?
A buffer solution is water mixed with a chemical to give it special properties in regards to pH (acidity). The chemical, known as a buffer agent, resists pH changes when exposed to acids and bases when properly mixed in a solution. This property makes it extremely useful in protecting sensitive equipment, dealing with chemical accidents, and even in balancing the internal processes of living things.
A buffer agent’s acid form (HA) is mixed with its conjugate base (A-) in water to make the solution. Once a balance exists between HA and A-, any other acids or bases added will be neutralized when they turn a portion of the buffer agent into either the HA or A- form. The change in the buffer agent initially causes little change in the solution’s pH. As the proportion of the agent’s HA and A- changes due to the addition of other acids and bases, the buffering capacity of the solution decreases. Eventually, a buffer agent can be used up to the point where it can no longer significantly resist pH changes, meaning it is no longer useful for this purpose.

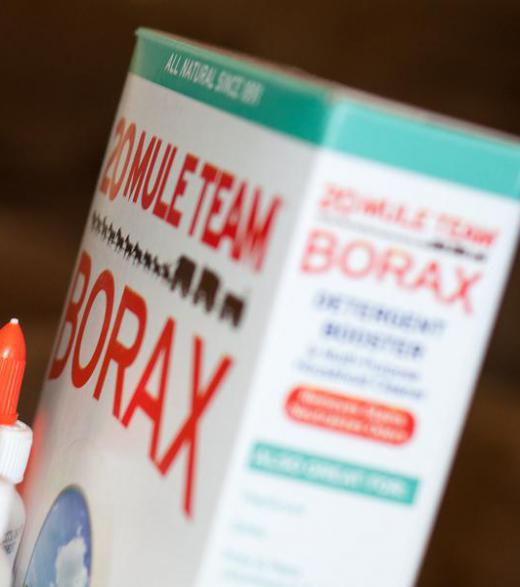
Buffer solutions and buffer agents are extremely common. The detergent borax, for instance, can be used to make a simple one. Many traditional antacid tablets are, in fact, buffer agents that work within the human body. Buffered aspirin is aspirin that has been treated with a buffering agent to help reduce pH changes to the aspirin when it is exposed to stomach acid. pH probes are protected from damage by being stored in these solutions.
In the human body, buffer agents play a vital role in both respiration and maintaining the body’s pH level. One of the most important agents in humans is bicarbonate. When carbon dioxide (CO2) mixes with water (H2O), carbonic acid is formed (H2CO3). Bicarbonate (HCO3-) is a dissociation of carbonic acid. Carbonic acid and bicarbonate form an equilibrium in the blood that helps the body adjust to pH changes.

Just like a buffer solution, the bicarbonate/carbonic acid equilibrium will no longer resist pH changes if their proportions are significantly altered. Excesses of carbonic acid are adjusted by forming carbon dioxide through the removal of water from carbonic acid; the carbon dioxide is then exhaled. Excessive amounts of bicarbonate can be reduced by inhaling more carbon dioxide, which is then changed into carbonic acid.
AS FEATURED ON:
AS FEATURED ON:












Discussion Comments
In my chemistry book, it says that a buffer solution can also be made by a weak base and its conjugate acid. Is that also true?
@turtlez - I think the most interesting part of the article is where it's talking about PH and the description of buffered aspirin. In this aspect, I think it is absolutely safe to say that the pool tablets used to balance the PH of a pool (normally chlorine tablets) are a buffer solution. In the end, they are helping to keep the pool at a safe PH balance in order to allow us to swim without burning our skin.
So does this mean that pool tablets are a buffer solution too?
There are several thousand buffer solutions available on the market today. The quickest thing I can think of (and something that almost everyone is familiar with) is the solution used in order to protect things like CDs and DVDs.
In some cases, people use spit (which is gross) as their buffer solution and then lightly rub it into a disc in order to prevent or repair scratches. While this isn't what most scientists think of when referring to a PH buffer solution, it's still a solution.
Post your comments