What Is an Azeotrope?
An azeotrope is a mixture of two or more chemicals in solution that cannot be separated by basic distillation processes because they share a common boiling point and vaporization point. When an azeotrope is heated or boiled, the chemicals all convert to a gaseous state in equal quantities, and they are sometimes referred to as constant boiling mixtures. Azeotropes can be difficult to make because the concentrations of each chemical must be precisely balanced, and their density, initial boiling point, and surface tension must be within a range that can be brought to a level of equilibrium in the solution.
The formation of azeotropic mixtures is more likely to occur if the precursor chemicals have boiling points that are close to each other. Chemicals with different electrical polarities or ionic charges are also more likely to be attracted to each other to form this type of mixture. Several types of solvents will naturally join into azeotropes due to these conditions, and they, therefore, have to be kept carefully separate during industrial processes.
In industry, the azeotrope has value because it is a mixture of chemicals in solution and not a compound where those chemicals exhibit strong molecular bonds that are not easily broken. This, therefore, makes it possible to design an azeotrope where the content consists of a flammable and a nonflammable chemical that overall demonstrates nonflammable characteristics. Since the chemicals don't separate during evaporation, spills will not result in hazardous fire conditions.

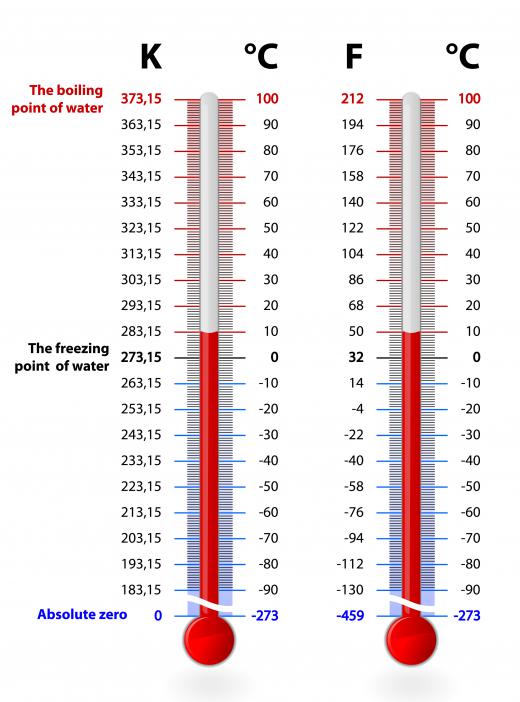
Flammable solvents are often part of azeotropic mixtures because they can be used and then the liquid distilled to recover the solvent and leave contaminates behind. The process avoids the risk of fire and air pollution by the solvent since it is bound to the nonflammable component of the azeotrope. The most common example of this type of solvent azeotrope is the binary ethanol-water azeotrope. Normally, water boils at 212° Fahrenheit (100° Celsius) and ethanol at 172.94° Fahrenheit (78.3° Celsius), but in a concentration of 95% ethanol and 5% water, they both boil at 172.76° Fahrenheit (78.2° Celsius). This type of mixture is also referred to as a minimum boiling azeotrope, due to the fact that the new boiling point for both constituents is below that of what it was originally.
A common methanol azeotrope that is a mixture of three chemicals, known as a ternary azeotrope, contains methanol, acetonitrite, and water in concentrations of 52%, 44%, and 4%, respectively. Combined, they have a boiling point between 149° to 158° Fahrenheit (65° to 70° Celsius), which is closest to the natural boiling point of methanol at 148.1° Fahrenheit (64.5° Celsius). This particular mixture is used in solvent recycling and the laboratory process of high-performance liquid chromatography (HPLC).
AS FEATURED ON:
AS FEATURED ON:









Discuss this Article
Post your comments