What is Fluorine?
 Mary McMahon
Mary McMahon
Fluorine is an extremely reactive and highly toxic gaseous element. In very small amounts, it is also a trace element necessary for the health of most organisms, although the line between enough and too much is very fine. The element and various compounds have a number of uses, ranging from the construction of nuclear bombs to additives in dental products which are designed to promote oral health. Pure fluorine is not generally available, since it is extremely dangerous, but fluorine compounds known as fluorides can be found in dental products and municipal water supplies, among other places.
The atomic number of fluorine is nine, and the element belongs to the halogen group. It is the lightest of the halogen gases, and the most reactive naturally occurring element, due to its electronegative configuration. On the periodic table of elements, fluorine is identified with the symbol F. The element was first isolated in 1886, at great peril to the scientists who did so.
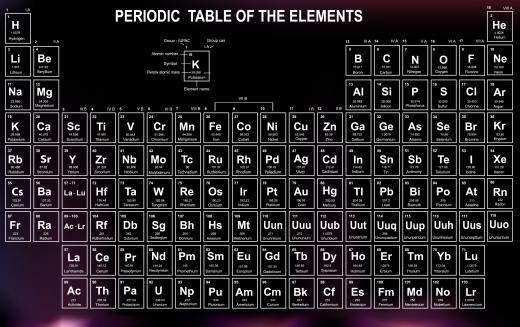
Pure fluorine is a pale yellow, highly corrosive, poisonous gas. It allegedly has a very pungent odor, although since inhalation of the gas is extremely dangerous, this is difficult to verify. It is essentially impossible to find pure fluorine in nature, since the highly reactive element readily bonds with any elements it is exposed to. It can also set off extremely explosive reactions which will continue until all of the fluorine has been consumed.

Compounds of the element have been used historically a flux agent, designed to make metal and slag more fluid during the smelting process. Fluorine is also a component in hydrofluoric acid, a compound used in etching, and the gas also has some medical uses. Dentists use sodium fluoride to help their patients establish strong, healthy teeth, and compounds of the gas are also used in anesthetics such as isoflurane. The element is also used in some antibiotics. In trace amounts for medical use, fluorine is combined with other elements so that it is nonreactive and safe.

Extreme caution should be used around pure fluorine and some fluorine compounds, because of their high toxicity. The element is extremely dangerous to inhale or touch, and contact with the skin or eyes should be carefully avoided. A high intake of fluorides can also be dangerous, leading to serious damage of bones and teeth, as well as corrosion of the intestinal tract. Products with fluorine in them such as fluoroquinolone antibiotics and fluorine based insecticides should be kept away from children and pets.
AS FEATURED ON:
AS FEATURED ON:















Discussion Comments
Sodium Fluoride is poison, plain and simple and they put it in our water supply and toothpaste to "make our teeth stronger". Do your research and stop using fluoride based toothpastes, unless they use calcium fluoride like Tom's of Maine.
Between what we drink, what we use in our toothpaste and what we absorb in our showers we are slowly poisoning ourselves to death or making ourselves very sick with autoimmune disorders.
i love it! well done you've really helped me in my science project! please tell me what the molecular structure is.
Post your comments