What Is Freezing Point Depression?
A freezing point depression is when the normal freezing point of a liquid is lowered beyond normal by the addition of a solute. Every liquid has a normal freezing point; for example, pure water freezes at 32 degrees Fahrenheit (F) (zero degrees Celsius, or C). The salt in sea water brings sea water to a lower freezing point than that of pure water; sea water can still freeze, however, the temperatures at which it does so are lower than waters from a pure water source. This fact is what enables the making of ice cream in ice cream freezers, when the metal canister in the middle is surrounded by rock salted ice. The melting of the ice by the rock salt allows the ice cream mix to lose heat into the surrounding ice water, and thereby freezes the ice cream.
Scientists have noted that the amount of solute added to a liquid is directly proportionate to the freezing point depression that is achieved. Not only do the molecules in a solution freeze at a lower temperature, but cooling down to a freezing point proceeds at a more moderate pace than in pure liquids. A solution’s freezing point depression capability is the basis for the use of salt or sand on icy roads in winter. The molecules of salt or sand, spread over roadways, mix with the ice and causes melt, which is particularly useful for the black ice that is so dangerous for vehicles to travel upon. Heavy spreading of salt can bring a freezing point depression down to as low as zero degrees F (-18 degrees C).
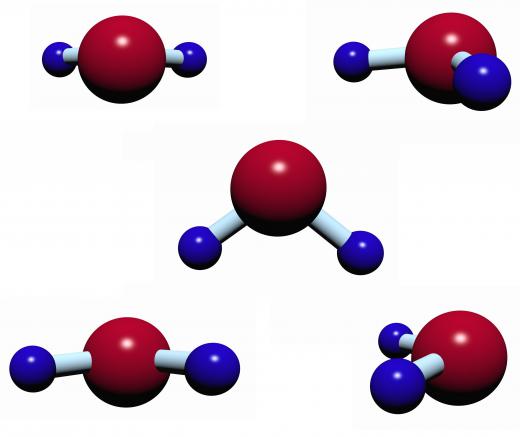
When salt is spread over ice, there is a chemical interchange between molecules. The surface molecules of ice begin escaping into melting water, and the surface molecules of water are trapped into the surface molecules of water. This shift between the molecules causes fluctuations in temperatures of each that gradually warm and melt the remaining ice as escaped molecules melt and speed up. The added mixture of salt introduces foreign matter molecules that do not easily pack into a solid and speed up liquification. For this reason, any foreign substance — alcohol, sugar, or salt — introduced would have the same effect, although salt is widely available and cheaper to use.

A freezing point depression is the intent of adding antifreeze to radiators in automotive vehicles. The addition of ethylene glycol, a main antifreeze ingredient, creates a solution with water. People in differing cold climates may use different percentages of antifreeze to water to reach differing depressions, however, when there is more antifreeze in the water than the reverse, the opposite effect happens and freezing points climb rather than depress. It is recommended that the sides of the antifreeze bottle be read to determine the best percentages to use in particular climates.
AS FEATURED ON:
AS FEATURED ON:









Discussion Comments
I want to know if blast freezing can kill all the bacteria present inside the food, and what temperature are enough to do it.
Post your comments