What Is Solvation?
In chemistry, when a substance known as a solute dissolves in another, known as a solvent, it is termed a solution. Solvation is a process where a chemical reaction occurs during this and both the solute and solvent combine to form weak covalent electron bonds. The process of solvation is closely related to dissolution and solubility, with the technical distinction that it creates a stable state in the solution where ionic charges of the solute and solvent cancel each other out to form an overall neutral charge.
Though the solvation model can also be applied to insoluble material where ion exchange processes occur on a solid surface, it is most often seen in aqueous solutions. It is considered an important process to understand in chemistry as most chemical reactions occur in a state of solution. In biology, essentially all biochemical reactions in the human body take place in a water-based solution as well, so solvation is even more important to understand there.
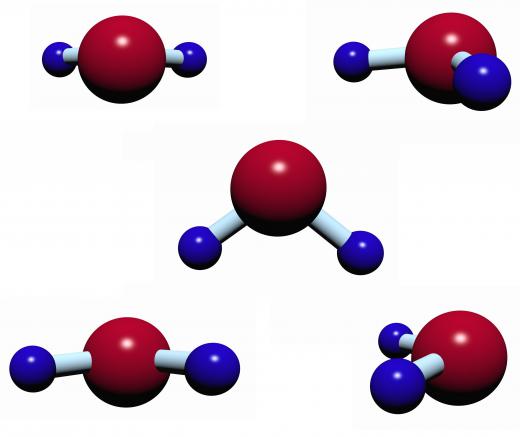
The common process of water solvation, also known as hydration, has a direct impact on the safety of city water supplies. Water that is purified for drinking and other uses by municipalities is not pure water, and, in fact, may end up with more dissolved solids in it due to hydration than it started out with. This is because water molecules generally contain enough ionic energy to break down the structure of crystal lattice ionic solids, such as sodium chloride.

The process of solvation only occurs with polar solvents, of which water is one. A polar solvent is a chemical where there is either a permanent separation of positive and negative charges or where the center for the charges is not balanced. Polar solvents are said to have high dielectric constants, or a strong ability to concentrate electric flux. Other examples of polar solvents include ethanol, butanol, and formic acid.
When the solvation process occurs, solvation energy levels also change. Energy is released when the solvent coordinates with the free ions, which is known as the energy of ligation. As the ions disperse in the solvent, the energy is bound up in a process known as energy of dispersion.
Some substances can release large amounts of energy as heat when solvation occurs in water, measured in kilogram-Joules/mol, and others release very little, depending on their molecular structure. The molar enthalpy, or heat content, of aluminum chloride, AlCl3, is -373.63 kJ/mol where that of sulfuric acid, H2SO4, is one-third that level at -95.28 kJ/mol, categorizing them as releasing high amounts of energy in a solvation of water. Examples of molecules that release small molar enthalpy rates include lithium nitrate, LiNO3, at -2.51 kJ/mol and potassium chloride, KCl, at -17.22 kJ/mol.
AS FEATURED ON:
AS FEATURED ON:









Discussion Comments
This article does a much better article describing what a solvent is in chemistry terms on how water does, indeed, act as one.
Post your comments