Which Material Has the Lowest Freezing Point?
 Michael Anissimov
Michael Anissimov
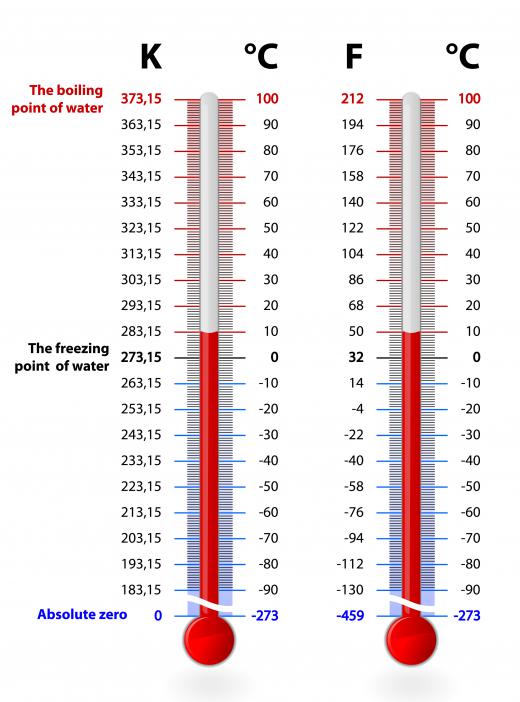
The material with the lowest freezing point is helium. Under typical pressures, it does not freeze at all, even at temperatures approaching absolute zero. The reasons why are dictated by quantum mechanics: the zero point energy of a helium system is too great to allow freezing. The zero point energy is the minimum energy a particle or system always has, no matter what. Helium is the only substance that does not have a freezing point under ambient pressure, no matter the temperature.
A freezing point for helium only exists under at least 25 atmospheres of pressure and a temperature of 1.15 K. These conditions have been created in a laboratory through evaporative cooling. The result is a colorless, highly compressible solid that is practically invisible. Solid helium is so difficult to see that layers of styrofoam are used just to tell where it is. The density of solid helium itself is only 66 times greater than air. By comparison, water is 1000 times more dense than air.
Helium was first liquefied in 1908 by Dutch physicist Heike Onnes, who cooled it to 1 degree Kelvin. Much to his surprise, further cooling did not cause it to reach its freezing point. It wasn’t until 18 years later, in 1926, that his student, Williem Keesom, was able to solidify helium by cooling it in a pressure chamber. Today, the liquefaction of helium is a vital step in extracting it from the earth and storing it.
Liquid helium is often used as a cryogenic cooling agent when liquid nitrogen isn’t enough. It must be kept under continuous high pressure and low temperature, otherwise it quickly expands and transitions to a gas. Solid helium does not have any practical applications outside of scientific research.
Some of helium’s most unusual properties can be coaxed out at temperatures close to absolute zero. At such temperatures, helium behaves as a superfluid, meaning it flows with zero measurable viscosity. It also has a tendency to creep up the walls of a container it is held in.
AS FEATURED ON:
AS FEATURED ON:








Discussion Comments
And, oddly, an extremely cold substance can effective cause a skin burn. Counter intuitive? Perhaps, but that's how it goes.
@Viranty - Based on my experience(s), I can answer that for you. I was once in a science lab, and I had to handle dry ice. Not aware of how cold it was, I picked it up (without wearing any gloves). My hands became severely frostbitten, and my skin was nearly damaged. Thankfully, it was taken care of pretty quickly (since my science teacher was nearby), but I learned my lesson from that day forward. Melonlity is right. Whether it's hot or cold, handling substances can be very dangerous, as they're not supposed to contact human skin. No matter the case, it's always good to be cautious.
@Melonity - I haven't had much experience handling cold substances, but what makes them so dangerous, and what happens if they come in contact with human skin?
@Melonlity - You took the words right out of my mouth. Though the article mainly discusses lowest freezing points, it's also important to note that cold substances can be extremely dangerous, sometimes even more so than hot substances. When handling these, not only is it a good idea to wear gloves, but it's an even better idea to pick up the material with tweezers or pliers. Most of us think we can handle the materials ourselves, but we all make mistakes, don't we? In the scientific realm, there's little room for error.
Good to know, and it's important to point out that handling liquid nitrogen or helium can be very dangerous. Horrible things happen when those substances come in contact with human skin.
Post your comments